|
Back to Blog
Molar mass of oxygen gas5/18/2023 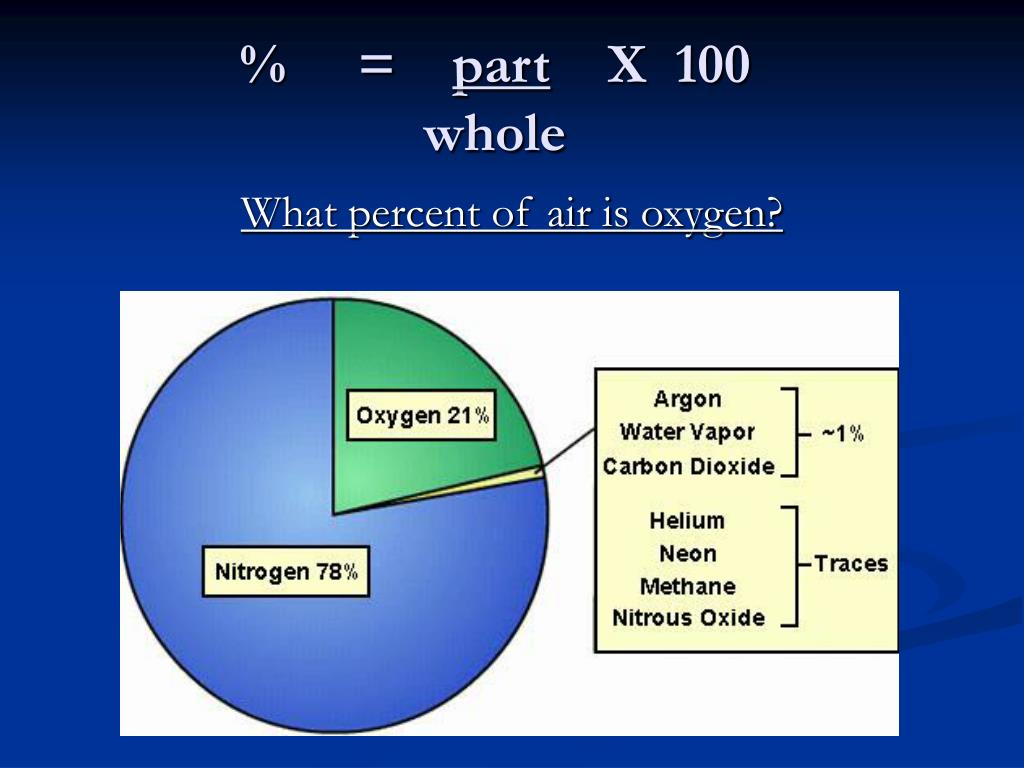
Please let us know how we can improve this web app. 
Related: Molecular weights of amino acids Weights of atoms and isotopes are from NIST article. This is exactly what we are doing when we calculate the relative molecular mass of a compound: Step 1: Use the chemical formula to determine how many atoms of each element are present in the compound. Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol. mass (water molecule) mass (oxygen atom) + 2 × mass (hydrogen atom) mass (water molecule) 16 + 2 × 1 16 + 2 18.(1 u is equal to 1/12 the mass of one atom of carbon-12) Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).For carbon dioxide, there is one carbon atom (weight 12) and two. To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations:ĭefinitions of molecular mass, molecular weight, molar mass and molar weight For oxygen, a molecule consists of two oxygen atoms, and has a weight of 2 x 16 32. Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Air is a mixture of several gases, where the two most dominant components in dry air are 21 vol oxygen and 78 vol nitrogen. 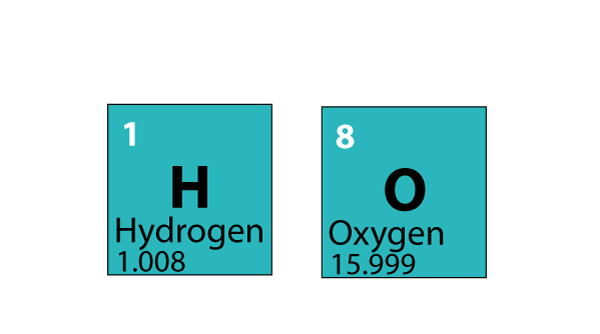
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
